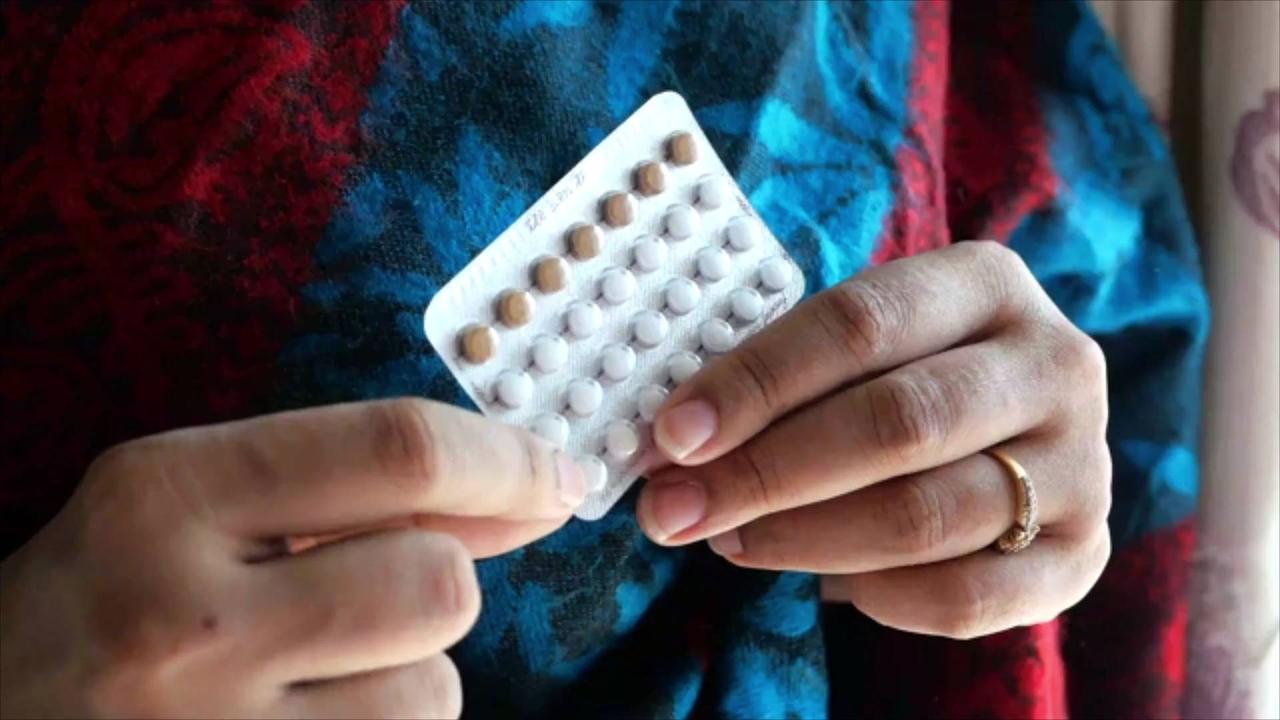
FDA Panel Backs , Over-the-Counter Birth Control Pill.
NPR reports that a panel of advisers has recommended that the Food and Drug Administration (FDA) approve the first over-the-counter birth control pill.
.
Pending the FDA's approval, Opill will be sold by Perrigo.
The progestin-only pill utilizes a synthetic version of the hormone to prevent pregnancy, while most other pills also contain estrogen.
I feel that the risk of unintended pregnancy is lower with this approach than any of the other available contraceptive approaches that women have access to without seeing a health care provider, Dr. Deborah Armstrong, a professor of oncology, gynecology and obstetrics at Johns Hopkins, via NPR.
I voted yes because the evidence demonstrates that the benefits clearly exceed the risks.
Opill has the potential to have a huge public health impact, Kathryn Curtis, a health scientist with the Centers for Disease Control and Prevention's division of reproductive health, via NPR.
NPR reports that the FDA questioned whether the company had provided enough evidence to convince the panel of the pill's safety without medical guidance.
One point the agency expressed concern about was if women with breast cancer would be aware that the pill would be dangerous for them to take.
Ultimately, the advisery panel concluded that women know enough about oral contraceptives to use the over-the-counter version safely and effectively.
I think this represents a landmark in our history of women's health.
Unwanted pregnancies can really derail a woman's life, and especially an adolescent's life, Dr. Margery Gass of the University of Cincinnati College of Medicine, via NPR.
So I'm very pleased that the FDA is seriously considering this.
And I look forward to it being on the market, Dr. Margery Gass of the University of Cincinnati College of Medicine, via NPR

